 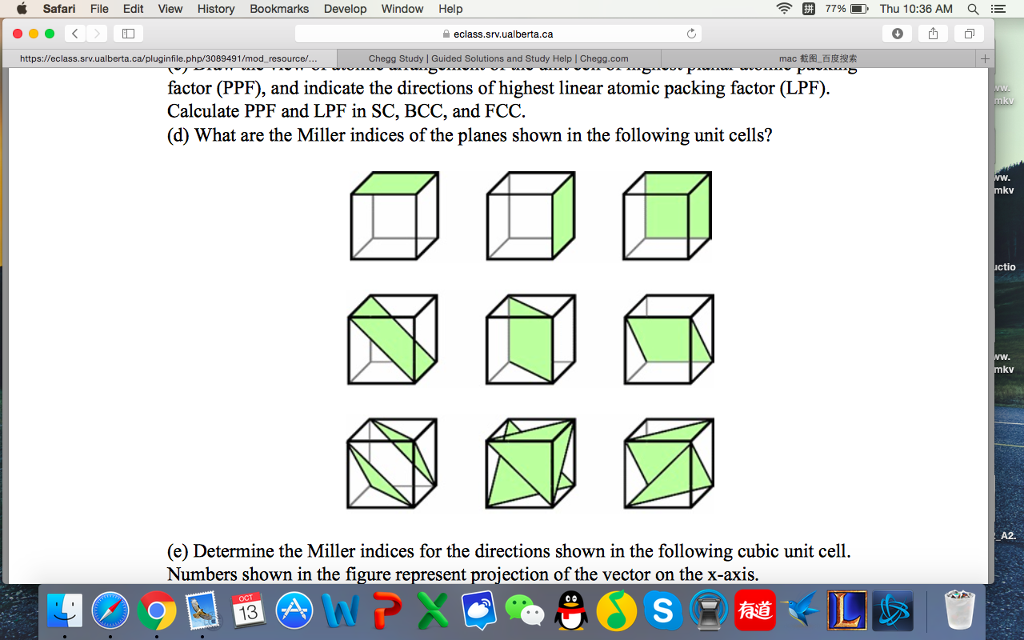
Why Things Break: Understanding the World by the Way It Comes Apart. Materials Science and Engineering: An Introduction 9th Edition, Wiley 9 edition (December 4, 2013), ISBN-13: 978-1118324578. Department of Energy, Material Science. DOE Fundamentals Handbook, Volume 2 and 2. January 1993. Department of Energy, Material Science. DOE Fundamentals Handbook, Volume 1 and 2. January 1993. This structure, along with its hexagonal relative (hcp), has the most efficient packing (74%). For example, metals with a high atomic packing factor will have a higher malleability or ductility, similar to how a road is smoother when the stones are closer together, allowing metal atoms to slide past one another more easily.įor example, in a fcc arrangement, a unit cell contains (8 corner atoms × ⅛) + (6 face atoms × ½) = 4 atoms. In materials science, the atomic packing factor of a unit cell explains many properties of materials. The radius of the spheres is taken to be the maximum value such that the atoms do not overlap. January 1993.īy convention, the APF is determined by assuming that atoms are rigid spheres. DOE Fundamentals Handbook, Volume 1 and 2. 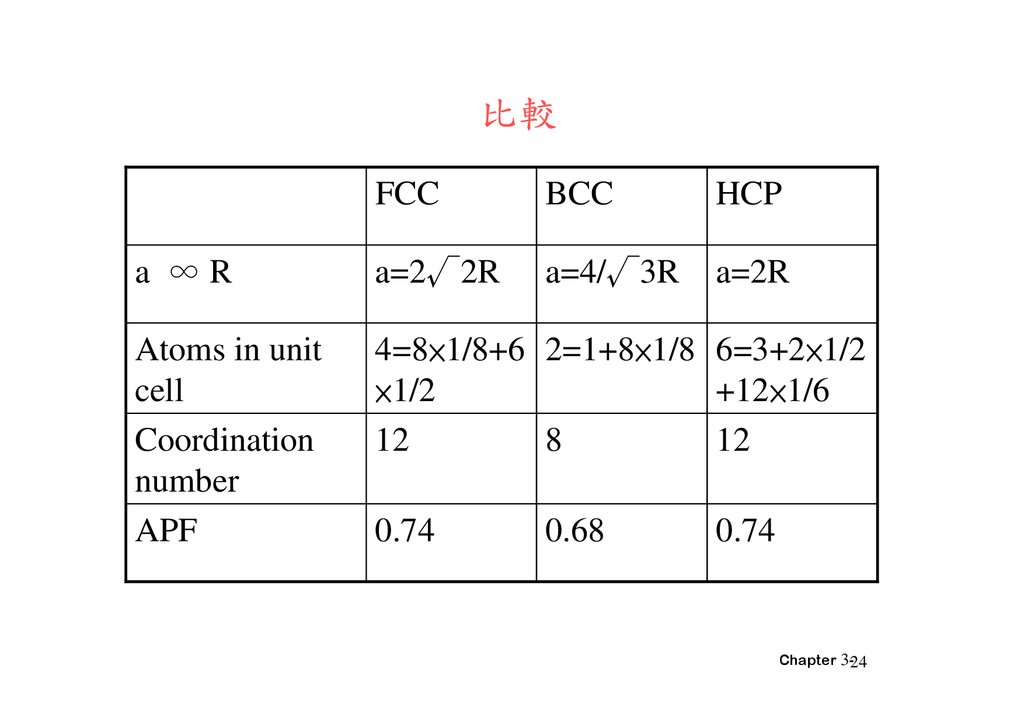
In crystallography, atomic packing factor (APF), packing efficiency or packing fraction is the sum of the sphere volumes of all atoms within a unit cell (assuming the atomic hard-sphere model) divided by the unit cell volume. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
